Industry and Regulatory Compliant Sampling Services
PrimusLabs offers trained samplers to meet your sampling needs including food & non-food contact surfaces, pre-harvest, raw & finished product samples, water samples, and more!

PrimusLabs Sampling Training
Confidently prepare you and your team with our Online Sampling Training. Let’s get started today!
The PrimusLabs team has developed self-guided sampling training for you. This program is designed to help you meet the training requirements in audit certification programs, specifically the PrimusGFS Certification Program. Upon successful completion, each trainee receives a certificate of completion that can be used as documentation for the PrimusGFS Audit.
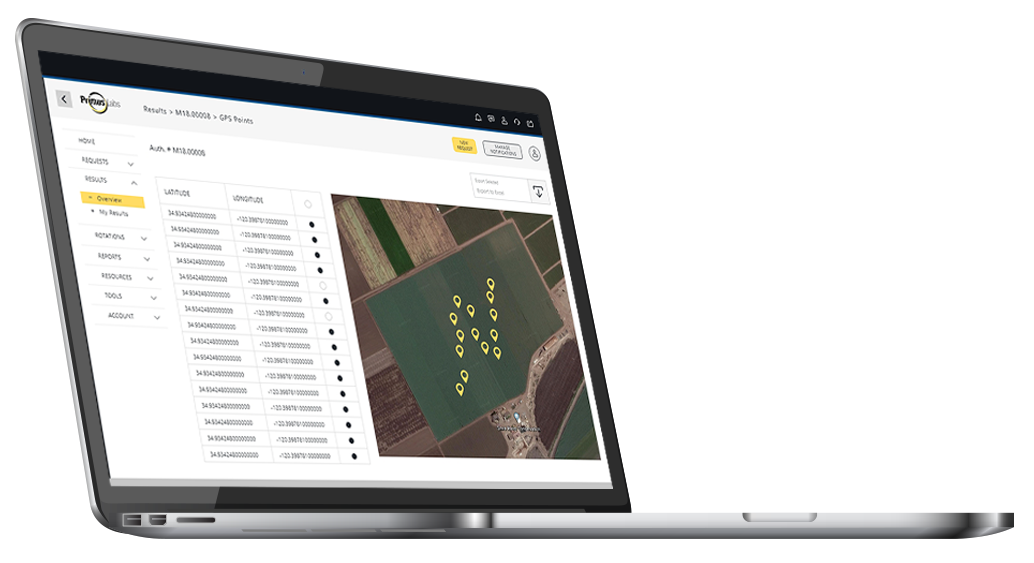
Field Sampling
PrimusLabs is an industry leader in field, pre-harvest testing. Samplers are trained in several protocols including LGMA and in-house SOPs, as well as client designed sampling plans.
If interested in specifying a particular sampling plan, please contact our sampling team.
GPS points are collected at each sample point throughout the field for enhanced chain-of-custody and confidence in sampling plan. GPS points are reported on all PrimusLabs reports.
Schedule a sampling request, request for analysis, or both online in a matter of minutes. Click ‘Begin a Request’ to explore all of your options for getting started.
Water Sampling
Sampling for regulatory or as part of a buyer requirement? PrimusLabs samplers collect samples from sources ranging from canals, irrigation systems, wells, and more.
If needed, all samplers are trained to follow LGMA guidelines for sampling.
Common tests on water include Generic E. coli (EC) and Total Coliform (TC). If looking to comply with FSMA’s Produce Rule requirements, our methods are included on the FDA’s list of Equivalent Testing Methodology for Agricultural Water.
GPS points are collected at each sample point for enhanced chain-of-custody. GPS points are reported on all PrimusLabs reports, allowing for additional analytics and trend analyses


Environmental Monitoring
PrimusLabs offers development of Rotation programs at no-charge to the customer. Simply provide an equipment list and sampling points you would like to target, and work with our staff to determine frequencies. Don’t know where to start, contact us to learn more!
Rotation development is available regardless of who samples. PrimusLabs offers sampling services including recurring events and one-off requests.
Or, if you prefer to sample and submit yourself, we are pleased to provide you with the rotations and corresponding sample point IDs.
Sample Point Trending and Data Analysis
With each sample point being unique, we are able to link them to the corresponding test result every time they are sampled. This allows for trending and analysis over time, and even further decision making capability.
FDA Detention Testing
FDA: Detention Without Physical Examination (DWPE)
The US Food & Drug Administration (FDA) has the ability to place companies on an Import Alert list when they find products in violation of regulations. Future samples imported into the U.S. are automatically detained (Detention without Physical Examination, or DWPE) and required to undergo specialized testing to be cleared.
Sampling and Procedures
Qualified third-party samplers must perform sampling for any detained product and each entry must follow a strict process for reporting of results to FDA.
Prior to scheduling sampling and testing, certain documents must be gathered including “FDA Notice of Action”, invoices, and specifics about commodity type, packaging information, and location of the product.
Testing
After sampling, the laboratories will test according to the appropriate protocol(s) and submit a comprehensive analytical report following FDA’s guidelines with extreme care to minimize delays caused by follow-up questions and re-submittals.
Contact our team today with any questions and to request a quote.

